Persistently elevated IgA antibodies to Mycoplasma pneumoniae in patients with internal carotid artery stenosis
Florian Daxböck 1Afshin Assadian 2
Thomas Watkins-Riedel 3
Ojan Assadian 4
1 Mödling, Austria
2 Department for Vascular and Endovascular Surgery, Wilhelminenspital Vienna, Vienna, Austria
3 Clinical Institute for Laboratory Medicine, Medical University Vienna, Vienna General Hospital, Vienna, Austria
4 Clinical Institute for Hospital Hygiene, Medical University Vienna, Vienna General Hospital, Vienna, Austria
Abstract
Background: It has been suggested that Mycoplasma pneumoniae may play a role in the development of atherosclerosis, but to date this association is still a matter of debate due to conflicting findings.
Methods: We have investigated the levels of specific IgA antibodies to M. pneumoniae in 91 patients with internal carotid artery (ICA) stenosis using a commercial kit (SeroMP™ IgA; Savyon Diagnostics, Israel; cut-off value: 20 binding units; BU). All patients underwent surgery for ICA stenosis. From each patient, the first serum sample (S1) was taken before surgery, and the second after an interval of 6 month (S2).
Results: The S1 seroprevalence was 18.7% (17/91). Thirteen of the 17 patients with positive S1 levels also remained positive after six month, whereby no decrease of IgA level was seen (median S1 level: 34 BU, range: 22–65 BU; median S2 level: 37 BU, range: 22–58 BU). Specifically, six of the patients showed an increased level after 6 months, and six a decrease, with the level remaining constant in one patient. In contrast, only 3 of the 74 S1 negative patients became positive for anti-M. pneumoniae IgA between the taking of the first and the second serum specimen (p<0.01). None of the assessed demographic factors or risk factors for atherosclerosis was associated with IgA seropositivity, neither were the degree CAVK or the degree of stenosis.
Conclusion: These findings cannot be explained throughout by the general seroprevalence, or by past respiratory tract infections with the pathogen, and therefore may suggest a role for M. pneumoniae in the development of atherosclerosis, since a chronic infection must be assumed.
Keywords
Mycoplasma pneumonia, IgA, atherosclerosis, stenosis, elevated antibodies, A. carotis interna
Introduction
Mycoplasma pneumoniae is an important cause of respiratory tract infections including community-acquired pneumonia [1]. It has been suggested that the pathogen may also play a role in the development of atherosclerosis. This hypothesis is supported by the ability of M. pneumoniae to cause extrapulmonary manifestations affecting the blood vessels [2], [3], [4]. Some recent studies found an association between M. pneumoniae infection and atherosclerosis [5], [6], [7]. However, others have presented discordant results, so that the role of M. pneumoniae in the pathogenesis of atherosclerosis must still be regarded as unknown [8], [9].
Serum IgA antibodies are a more reliable indicator for acute M. pneumoniae infection than IgM, because specific IgM are not mounted regularly in elderly patients [10], [11]. Importantly, the serum level of specific IgA declines to normal values earlier after acute infection than the level of specific IgM, with a significant decrease 60 days after the onset of disease [12]. Therefore, persistently elevated IgA serum levels may indicate chronic infection. The aim of this study was to investigate the role of M. pneumoniae in the development atherosclerosis by repeated testing of specific IgA over a period of 6 month.
Material and methods
Ninety-one consecutive patients with symptomatic and asymptomatic internal carotid artery (ICA) stenosis >70%, who were admitted for surgery March and July 2004 were enrolled in this study. Fifty-three patients were male and 38 were female. The age ranged from 41 to 86 years (median: 69 years). Thirty-nine patients (43%) had asymptomatic ICA stenosis, 32 patients (35%) had transient ischaemic episodes and 20 patients (22%) were operated on for non-disabling stroke.
All patients were planned for routine eversion endarterectomy (EEA) of the ICA under regional anaesthesia. As anaesthetic agent 1% lidocaine was used for a combined superficial and deep cervical block. Symptomatic patients with stroke had an interval of less than 6 weeks prior to surgery; symptomatic patients with transient hemispheric and non-hemispheric symptoms were all operated on within 3 weeks after presentation. For all patients, the following data were assessed: Age, sex, clinical stage of carotid artery disease (CAD) (right/left), degree of stenosis (right/left), body mass index (BMI), alcohol consumption (gram/week), nicotine (cigarettes/day), diabetes mellitus, cholesterol, triglycerides and homocysteine.
Patients on antihypertensive medication or a blood pressure of ≥140/75 mmHg on repeat measurements were characterized hypertensive.
Maximum ICA stenosis was assessed by magnetic resonance angiography and Duplex and stenosis of ≥70% were an indication for surgery.
Serum was obtained by venipuncture of the antecubital vein using standard tubes (Vacuette EDTA Tubes, Greiner Bio-One, Kremsmuenster, Austria) and centrifugation 10 minutes at 2,000 G. Serum was stored at –80°C until use. The first serum specimen (S1) was taken before surgery, and the second after an interval of 6 months (S2). Specific IgA were measured by the SeroMP™ IgA kit (Savyon Diagnostics, Ashdod, Israel) according to the manufacturer’s recommendations. As indicated by the manufacturer values >20 BU (Binding Units) were considered positive.
All patients gave written informed consent. The study was approved by the local ethics committee.
Statistical analysis was performed by Chi squared test (Yates correction), using EpiInfo 2002™ (CDC, Atlanta, GA, USA). A p-value of <0.05 was considered significant.
Results
The IgA seroprevalence at admission (S1) was 18.7% (17 of 91 patients). In these patients, the median S1 value was 35 BU (range: 22–58 BU). Thirteen of these 17 patients were also tested positive for anti-M. pneumoniae IgA after a period of 6 month (median: 36 BU; range: 22–57 BU). Among the 74 patients with negative S1 results, only 3 showed a positive result after 6 months (p<0.01). The ELISA results and basic clinical data of the 18 patients with ≥1 result >20 BU are shown by Table 1 [Tab. 1].
Table 1: Results of IgA ELISA and clinical data in 18 patients who were tested positive (>20 binding units; BU) on at least one occasion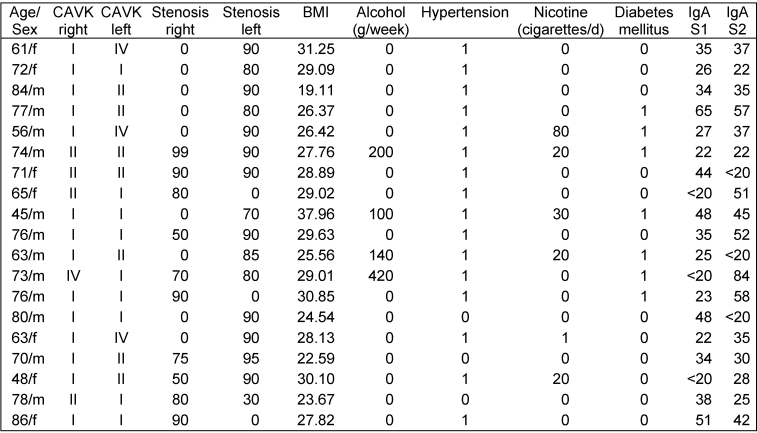
None of the assessed demographic factors or risk factors for atherosclerosis was associated with IgA seropositivity, neither were the degree CAVK or the degree of stenosis.
Discussion
In this study we found a considerable seroprevalence of IgA antibodies to M. pneumoniae in patients with internal carotid artery stenosis. However, the more remarkable finding was that in the majority of our patients IgA remained elevated at essentially constant levels through a period of at least 6 month. In contrast, few patients (3) developed specific IgA between the taking of the first and the second serum specimen.
We have focused on IgA determination in the present study, because specific IgM formation may be lacking in the elderly [10]. IgG antibodies, on the other hand, remain elevated for longtime after infection, which results in a high IgG seroprevalence, that can cause interpretation difficulties [1].
Determination of specific IgA is a suitable tool for the diagnosis of M. pneumoniae infection [11]. It has been shown that after infection specific IgA decline to normal values (i.e. levels below the cut-off value of commercially available tests) earlier than specific IgM [12]. However, IgA-based diagnosis is less established than IgM- or IgG-based diagnosis, and persistently elevated IgA as indicator of chronic infection, although biologically plausible, has to be interpreted with caution. It has to be mentioned that differing figures concerning the seroprevalence of IgA antibodies to M. pneumoniae in elderly patients have been published, with seroprevalences up to 79% [8], [13]. However, the applied assay and the respective cut-off level for positivity is obviously important in this context. By the assay applied in the present study, we have found a seroprevalence of 13% (>20 BU) and no value >30 BU among 46 healthy subjects [11].
A certain variance of ELISA results has to be considered. Nevertheless, a significant trend to a decrease in antibody level would be expected if the elevated IgA levels in our patients should simply reflect past respiratory tract infection. It is notable that in the 13 patients with persistently high IgA levels, 6 showed an increasing and 6 a decreasing IgA level, with the level in one patient remaining constant (Table 1 [Tab. 1]).
An important study negotiating a potential role of M. pneumoniae in the development of atherosclerosis has been provided by Maraha et al., who failed to detect the pathogen in the great majority of 103 atherectomy specimens and degenerative heart valve specimens [9]. However, it should be considered that some mycoplasma-associated diseases, including severe pneumonia and neurological manifestations, are immunologically mediated and require the presence of the pathogen only in an early stage of the disease [14], [15], [16]. Therefore, serological methods may be advantageous to explore the association between M. pneumoniae infection and atherosclerosis.
In conclusion, we have applied a new serological approach to investigate the role of M. pneumoniae in the development of atherosclerosis. The results can not be explained throughout by the general seroprevalence, or by past respiratory tract infections with the pathogen, so that they suggest a role for M. pneumoniae in the development of atherosclerosis. However, it has to be considered that the association between chronic mycoplasma infection and persistently elevated IgA antibodies is not yet proven by large-scale epidemiological investigations. Future studies on the potential role of M. pneumoniae in the development of atherosclerosis are warranted.
Notes
Conflicts of interest
The authors declare that they have no competing interests.
References
[1] Waites KB, Talkington DF. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev. 2004;17(4):697-728. DOI: 10.1128/CMR.17.4.697-728.2004[2] Antachopoulos C, Liakopoulou T, Palamidou F, Papathanassiou D, et al. Posterior cerebral artery occlusion associated with Mycoplasma pneumoniae infection. J Child Neurol. 2002;17(1):55-7. DOI: 10.1177/088307380201700115
[3] Fu M, Wong KS, Lam WW, et al. Middle cerebral artery occlusion after recent Mycoplasma pneumoniae infection. J Neurol Sci. 1998;157(1):113-5. DOI: 10.1016/S0022-510X(98)00074-4
[4] Dowd AB, Grace R, Rees WD. Cerebral infarction associated with Mycoplasma pneumoniae infection. Lancet. 1987;330(8558):567. DOI: 10.1016/S0140-6736(87)92950-3 (Originally published as Volume 2, Issue 8558.)
[5] Reunanen A, Roivainen M, Kleemola M. Increased titre of antibodies to Mycoplasma pneumoniae may be associated with coronary heart disease. Atherosclerosis. 2005;180(1):209-10. DOI: 10.1016/j.atherosclerosis.2004.12.032
[6] Reunanen A, Roivainen M, Kleemola M, Saikku P, et al. Enterovirus, mycoplasma and other infections as predictors for myocardial infarction. J Intern Med. 2002;252(5):421-9. DOI: 10.1046/j.1365-2796.2002.01052.x
[7] Momiyama Y, Ohmori R, Taniguchi H, et al. Association of Mycoplasma pneumoniae infection with coronary artery disease and its interaction with chlamydial infection. Atherosclerosis. 2004;176(1):139-44. DOI: 10.1016/j.atherosclerosis.2004.04.019
[8] Ngeh J, Gupta S, Goodbourn C, et al. Mycoplasma pneumoniae in elderly patients with stroke. A case-control study on the seroprevalence of M. pneumoniae in elderly patients with acute cerebrovascular disease – the M-PEPS Study. Cerebrovasc Dis. 2004;17(4):314-9. DOI: 10.1159/000077342
[9] Maraha B, van Der Zee A, Bergmans AM, Pan M, et al. Is Mycoplasma pneumoniae associated with vascular disease? J Clin Microbiol. 2000;38(2):935-6.
[10] Sillis M. The limitations of IgM assays in the serological diagnosis of Mycoplasma pneumoniae infections. J Med Microbiol. 1990;33(4):253-8. DOI: 10.1099/00222615-33-4-253
[11] Watkins-Riedel T, Stanek G, Daxboeck F. Comparison of SeroMP IgA with four other commercial assays for serodiagnosis of Mycoplasma pneumoniae pneumonia. Diagn Microbiol Infect Dis. 2001;40(1-2):21-5. DOI: 10.1016/S0732-8893(01)00250-4
[12] Granstrom M, Holme T, Sjogren AM, et al. The role of IgA determination by ELISA in the early serodiagnosis of Mycoplasma pneumoniae infection, in relation to IgG and mu-capture IgM methods. J Med Microbiol. 1994;40(4):288-92. DOI: 10.1099/00222615-40-4-288
[13] Tuuminen T, Varjo S, Ingman H, et al. Prevalence of Chlamydia pneumoniae and Mycoplasma pneumoniae immunoglobulin G and A antibodies in a healthy Finnish population as analyzed by quantitative enzyme immunoassays. Clin Diagn Lab Immunol. 2000;7(5):734-8.
[14] Daxbock F, Brunner G, Popper H, Krause R, et al. A case of lung transplantation following Mycoplasma pneumoniae infection. Eur J Clin Microbiol Infect Dis. 2002;21(4):318-22. DOI: 10.1007/s10096-002-0698-0
[15] Radisic M, Torn A, Gutierrez P, et al. Severe acute lung injury caused by Mycoplasma pneumoniae: potential role for steroid pulses in treatment. Clin Infect Dis. 2000;31(6):1507-11. DOI: 10.1086/317498
[16] Jacobs BC, Rothbarth PH, van der Meche FG, Herbrink P, et al. The spectrum of antecedent infections in Guillain-Barre syndrome: a case-control study. Neurology. 1998;51(4):1110-5.




