Angiogenesis, cell differentiation and cell survival in tissue engineering and cancer research
Daniel Johannes Tilkorn 11 Klinik für Plastische, Rekonstruktive und Ästhetische Chirurgie, Handchirurgie, Alfried Krupp Krankenhaus, Essen, Germany
Abstract
Recent medical advances lead to a growing demand for tissue engineering and regenerative medicine in the future. Tissue engineering and regenerative medicine aim to create substitute tissue or restore lost or impaired tissue by combining biological science with engineering techniques, whereas cancer research faces the challenge to identify and hinder aberrant and uncontrolled cell growth.
These two seemingly opposing fields of research share fundamental communalities.
This review focuses on the shared underlying biological processes. Exploring these mechanisms of tissue growth and homeostasis from different angles will allow for creative novel approaches for both areas of research.
Introduction
The increase in life expectance and the ever growing medical progress allows an increased number of patients to survive cancer, severe trauma as well as chronic diseases, leading to a higher demand in substitute tissue to replace lost or diseased tissue or organs.
At present, these patients depend upon reconstructive procedures, requiring the transplantation of autologous or allogenic tissue. These procedures present some specific drawbacks such as donor site morbidity or adverse immunoreactions.
Tissue engineering and regenerative medicine combine engineering and biological principals, which aim to regenerate or create new tissue and organs using autologous cells to circumvent immunological reactions. In addition this tissue may also be used for diagnostic and research purposes.
It represents a great challenge for tissue engineering disciplines to create an ideal environment for cells to grow, survive and differentiate.
Stem or progenitor cells, extracellular matrices and a functional vascular network are essential prerequisites for successful generation of substitute tissue.
It is crucial for the development of clinically valuable tissue to enhance cell survival, differentiation as well as proliferation and to allow cell interaction, apoptosis and angiogenesis [1], [2], [3].
These biological processes mentioned above are oftentimes affected in tumor genesis, in which aberrant cell growth, unhindered cell proliferation, altered cell signaling and resistance towards apoptosis promote malignant growth [4].
In contrast to tissue engineering research the identification and inhibition of these underlying processes represent the greatest challenge for cancer research.
Even though tissue engineering and cancer research are seemingly opposing fields of research, there exist great similarity in regards to the underlying biological processes.
The unique properties of stem and progenitor cells and their role in tissue and organ regeneration bare great potential for engineering tissue constructs. On the other hand emerging evidence implicate stem cells and progenitor cells as the source of oncogenic transformation [5], [6], [7], [8], [9], [10]. A functional vascular network and successful neoangiogenesis is the essential basis for cell survival in vivo. Transplanted cells within tissue engineering constructs will not survive unless they are in close proximity or have access to a functional vascular network for nutritional and oxygen supply and waste deposition [11].
The same holds true for rapid growing malignant tumors.
The first vascular growth factors were discovered within well-vascularized tumors [12] and are now widely explored in the tissue engineering setting to enhance neoangiogenesis and cell survival in tissue constructs.
Cell signaling and cell differentiation play a significant and obvious role for cell based regenerative medicine [13], [14], [15] and have recently been introduced into the cancer setting.
Hence a profound knowledge of the fundamental biological process and interaction, which influence cell survival, differentiation and proliferation as well as apoptosis, is of great importance for both fields of research.
To further elucidate these fundamental communalities, a set of research experiments was conducted with a special focus on cell survival and differentiation both in tissue engineering constructs and tumor models.
The initial experiments assessed the role of angiogenesis, differentiation and cell survival in the tissue engineering setting. The gained knowledge was then translated into the cancer setting.
Tissue engineering experiments
In order to evaluate the influence of various matrix materials on the angiogenic process, two different matrices, a lacto polymer and a PEGT/PBT copolymer were analyzed in an in vivo skin fold chamber in a mouse model. Intravital fluorescent microscopy allowed for a continuous analysis of the angiogenic process and the study of changes in microcirculation in vivo [16] (Figure 1 [Fig. 1]).
Figure 1: Dorsal skin fold chamber for intravital microscopy in situ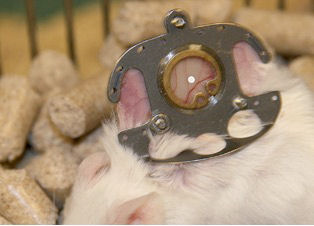
The angiogenic process was detected as early as on the first day with a peak on the seventh day when a functional vascular network has been formed.
The importance of the vasculature on the survival of transplanted cells was verified for different cell types, myoblasts [17], thymocytes [18], osteoblasts and dental pulp cells [19], [20], using a tissue engineering model with intrinsic vascular supply. For this purpose the tissue engineering chamber was centered around superficial epigastric vessels as vascular pedicle in mice (Figure 2 [Fig. 2], Figure 3 [Fig. 3]). Further we could show the time relationship between angiogenesis and cell survival following cell implantation within a tissue engineering model in the groin of inbreed rats. An arteriovenous shunt was placed in a tissue engineering chamber on day 1 of the experiment. Myoblasts were inserted into the chamber on day 1, day 4 or day 7 according to experimental protocol. A marked increase in cell survival was detected when cells were implanted on day 4 or even more for cells implanted on the seventh day after establishing the tissue engineering construct, when a functional vascular network has been formed [17]. In order to detect cell innate factors able to improve cell survival under hypoxic conditions, myoblasts were preconditioned with either hypoxia, nitric oxide, hypoglycemia or hyperthermia prior to the implantation into the tissue engineering chamber (Figure 4 [Fig. 4]). Exposure to a sublethal dose of cell stress (preconditioning) significantly enhanced cell survival and led to a higher vascular density as well as cell differentiation [21] (Figure 5 [Fig. 5]).
Figure 2: Tissue engineering chamber with intrinsic vascularisation centred around the superficial epigastic vessels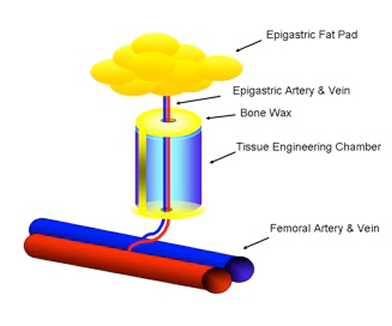
Figure 3: De novo generated tissue within the tissue engineering chamber model in mice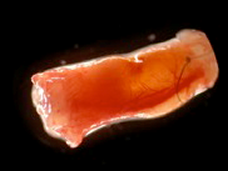
Figure 4: Experimental preconditioning regiment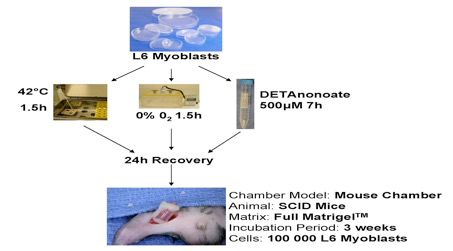
Figure 5: Differentiated myoblasts following hypoxic preconditioning and in vivo implantation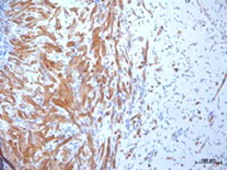
In addition it was shown that adding dentin fragments as differentiation clues to the tissue engineering model apart from initiating cell differentiation, also positively influenced survival dental pulp stem cells [19], [20].
Translation into cancer research
The knowledge gained from the tissue engineering experiments was transferred in the cancer setting to improve the reproducibility and comparability of sarcoma models.
An in vivo model with intrinsic vascular supply was developed in order to grow human sarcomas in mice and was compared to the commonly used subcutaneous implantation of tumor cells.
Tumors which had been grown in the model with intrinsic vascularization closely resembled the human primary tumor and allowed an improved evaluation of tumor angiogenesis and tumor matrix interaction [22], [23].
Finally tumor cells were shown to alter stromal cell differentiation in a chemokine fashion by adding cell free sarcoma media to preadipocytes in vitro [24].
Conclusion
In conclusion a set of experiments demonstrated the importance of the biological process of angiogenesis, cell differentiation and cell survival both for tissue engineering and cancer research. Cellular pathways and mechanisms directing cell survival and function to successful tissue engineering outcomes are often examined in the cancer setting and vice versa. Acquiring insights in the biological processes to control the growth, differentiation and growth inhibition of a tissue engineered construct are challenges at the forefront of tissue engineering. Knowledge gained from cancer research have led to many novel insights including the identification of specific factors and signaling pathways that are aberrantly activated in the tumor. Elucidating these fundamental mechanisms of cancer development and tissue growth, homeostasis and regeneration will yield novel approaches for both areas of research. Many fundamental mechanisms of cancer development as well as tissue regeneration conjoin these opposing fields of research. Shining light on these mechanisms of tissue growth and homeostasis from different angles will allow for creative novel approaches for both areas of research [4].
Notes
Competing interests
The author confirms that there are no financial or non-financial conflicts of interest in regards to this article.
References
[1] Langer R, Vacanti JP. Tissue engineering. Science. 1993 May;260(5110):920-6. DOI: 10.1126/science.8493529[2] Vacanti CA. The history of tissue engineering. J Cell Mol Med. 2006 Jul-Sep;10(3):569-76. DOI: 10.1111/j.1582-4934.2006.tb00421.x
[3] Vacanti CA. History of tissue engineering and a glimpse into its future. Tissue Eng. 2006 May;12(5):1137-42. DOI: 10.1089/ten.2006.12.1137
[4] Tilkorn DJ, Lokmic Z, Chaffer CL, Mitchell GM, Morrison WA, Thompson EW. Disparate companions: tissue engineering meets cancer research. Cells Tissues Organs (Print). 2010;192(3):141-57. DOI: 10.1159/000308892
[5] Gupta PB, Chaffer CL, Weinberg RA. Cancer stem cells: mirage or reality? Nat Med. 2009 Sep;15(9):1010-2. DOI: 10.1038/nm0909-1010
[6] Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007 Oct;449(7162):557-63. DOI: 10.1038/nature06188
[7] Weinberg R, Fisher DE, Rich J. Dynamic and transient cancer stem cells nurture melanoma. Nat Med. 2010 Jul;16(7):758. DOI: 10.1038/nm0710-758
[8] Chaffer CL, Weinberg RA. A perspective on cancer cell metastasis. Science. 2011 Mar;331(6024):1559-64. DOI: 10.1126/science.1203543
[9] Chaffer CL, Weinberg RA. Cancer cell of origin: spotlight on luminal progenitors. Cell Stem Cell. 2010 Sep;7(3):271-2. DOI: 10.1016/j.stem.2010.08.008
[10] Fletcher CD. The evolving classification of soft tissue tumours: an update based on the new WHO classification. Histopathology. 2006 Jan;48(1):3-12. DOI: 10.1111/j.1365-2559.2005.02284.x
[11] Folkman J, Hochberg M. Self-regulation of growth in three dimensions. J Exp Med. 1973 Oct;138(4):745-53. DOI: 10.1084/jem.138.4.745
[12] Ferrara N. Role of vascular endothelial growth factor in physiologic and pathologic angiogenesis: therapeutic implications. Semin Oncol. 2002 Dec;29(6 Suppl 16):10-4. DOI: 10.1053/sonc.2002.37264
[13] Friedenstein AJ. Precursor cells of mechanocytes. Int Rev Cytol. 1976;47:327-59. DOI: 10.1016/S0074-7696(08)60092-3
[14] Bianco P, Riminucci M, Gronthos S, Robey PG. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells. 2001;19(3):180-92. DOI: 10.1634/stemcells.19-3-180
[15] Owen M, Friedenstein AJ. Stromal stem cells: marrow-derived osteogenic precursors. Ciba Found Symp. 1988;136:42-60.
[16] Ring A, Goertz O, Al-Benna S, Ottomann C, Langer S, Steinstraesser L, Schmitz I, Tilkorn D. Accelerated angiogenic induction and vascular integration in a novel synthetic scaffolding matrix for tissue replacement. Int J Artif Organs. 2010 Dec;33(12):877-84.
[17] Tilkorn DJ, Bedogni A, Keramidaris E, Han X, Palmer JA, Dingle AM, Cowling BS, Williams MD, McKay SM, Pepe L, Deftereos A, Morrison WA, Penington AJ, Mitchell GM. Implanted myoblast survival is dependent on the degree of vascularization in a novel delayed implantation/prevascularization tissue engineering model. Tissue Eng Part A. 2010 Jan;16(1):165-78. DOI: 10.1089/ten.TEA.2009.0075
[18] Seach N, Mattesich M, Abberton K, Matsuda K, Tilkorn DJ, Rophael J, Boyd RL, Morrison WA. Vascularized tissue engineering mouse chamber model supports thymopoiesis of ectopic thymus tissue grafts. Tissue Eng Part C Methods. 2010 Jun;16(3):543-51. DOI: 10.1089/ten.TEC.2009.0135
[19] Srisuwan T, Tilkorn DJ, Al-Benna S, Abberton K, Messer HH, Thompson EW. Revascularization and tissue regeneration of an empty root canal space is enhanced by a direct blood supply and stem cells. Dent Traumatol. 2013 Apr;29(2):84-91. DOI: 10.1111/j.1600-9657.2012.01136.x
[20] Srisuwan T, Tilkorn DJ, Al-Benna S, Vashi A, Penington A, Messer HH, Abberton KM, Thompson EW. Survival of rat functional dental pulp cells in vascularized tissue engineering chambers. Tissue Cell. 2012 Apr;44(2):111-21. DOI: 10.1016/j.tice.2011.12.003
[21] Tilkorn DJ, Davies EM, Keramidaris E, Dingle AM, Gerrand YW, Taylor CJ, Han XL, Palmer JA, Penington AJ, Mitchell CA, Morrison WA, Dusting GJ, Mitchell GM. The in vitro preconditioning of myoblasts to enhance subsequent survival in an in vivo tissue engineering chamber model. Biomaterials. 2012 May;33(15):3868-79. DOI: 10.1016/j.biomaterials.2012.02.006
[22] Tilkorn D, Daigeler A, Stricker I, Schaffran A, Schmitz I, Steinstraesser L, Hauser J, Ring A, Steinau HU, Al-Benna S. Establishing efficient xenograft models with intrinsic vascularisation for growing primary human low-grade sarcomas. Anticancer Res. 2011 Dec;31(12):4061-6.
[23] Tilkorn DJ, Daigeler A, Hauser J, Ring A, Stricker I, Schmitz I, Steinstraesser L, Steinau HU, Al-Benna S. A novel xenograft model with intrinsic vascularisation for growing undifferentiated pleomorphic sarcoma NOS in mice. J Cancer Res Clin Oncol. 2012 May;138(5):877-84. DOI: 10.1007/s00432-012-1163-9
[24] Tilkorn DJ, Al-Benna S, Hauser J, Ring A, Steinau HU, Tannapfel A, Stricker I. Sarcoma cells induce alteration in adipogenic differentiation. Anticancer Res. 2012 Apr;32(4):1167-73.




